
American Society of Clinical Oncology ASCO releases the data on Polyoxidonium use in cancer therapy
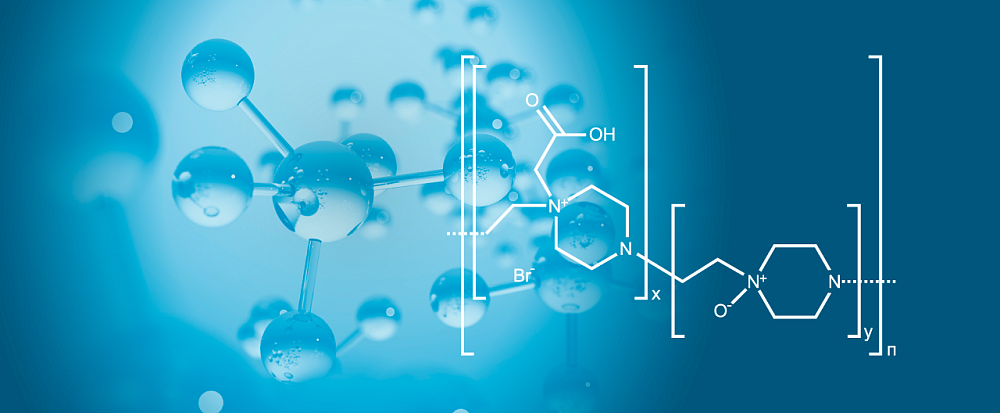
American Society of Clinical Oncology (ASCO), one of the most authoritative scientific oncology communities, has published data received by Russian researchers on the use of the original medicinal product Polyoxidonium (Azoximer bromide) produced by Petrovax Pharm in the therapy of cancer.
The results of the retrospective study were posted on the society's website as part of the annual international conference, which is held this year online on June 4–8.
During the study, the data of 1,391 patients with melanoma and 243 patients with soft tissue sarcomas treated at the NMRC of Oncology named after N.N.Petrov, St. Petersburg, were analyzed. In the group of patients observed, 42 patients with melanoma and 25 patients with soft tissue sarcomas were administered with Azoximer bromide (AB) in addition to the standard of care.
The researchers concluded that the addition of Аzoximer bromide (AB) to the standard of care for melanoma and soft tissue sarcomas is an independent favorable factor positively influencing the disease prognosis.
Polyoxidonium (Аzoximer bromide) was originally developed by the Petrovax Pharm. Currently, the company executes R&D program and invests significant resources for expanding the evidential base for proprietary medicines including activities on the global level. The evaluation of Polyoxidonium for cancer therapy is a part of the in-house research strategy program for the medicinal product use in socially-important therapy.
Reference:
The American Society of Clinical Oncology established in 1964, brings together more than 45,000 experts in oncology from over 150 countries. The main goal of ASCO is to combat cancer diseases by means of research and education activities. The clinical guidelines issued by ASCO are the generally accepted standard of care for cancer patients oriented by most regulatory authorities in the development of locally accepted clinical recommendations.
Petrovax and Government of the Moscow Region Sign a Cooperation Agreement to Improve the Investment Climate and Economy of the Region


